Research
Functional Analysis of BAF complexes
Created in BioRender.
BAF complex diversity
BAF complexes come in many different forms. In addition to the previously recognized canonical BAF (cBAF) and Polybromo BAF (PBAF) complexes, we discovered a new variant of the BAF complex called the non-canonical or ncBAF complex, which incorporates the bromodomain protein BRD9. By profiling subunits unique to cBAF, ncBAF, or PBAF, we have shown that BAF variants have shared and independent binding and function. The genomic localization of ncBAF complexes is regulated by the binding of the BRD9 bromodomain to acetylated residues. Given that many BAF subunits contain protein domains that interface with chromatin, we are performing biochemical and molecular biology analysis to elucidate the logic of complex targeting. Taking advantage of CRISPR/Cas9 base editing, we are interrogating another source of regulation of which we know very little – post-translational modifications (PTMs) to BAF complex subunits – in BAF complex binding and function.
BAF complex mechanism of action
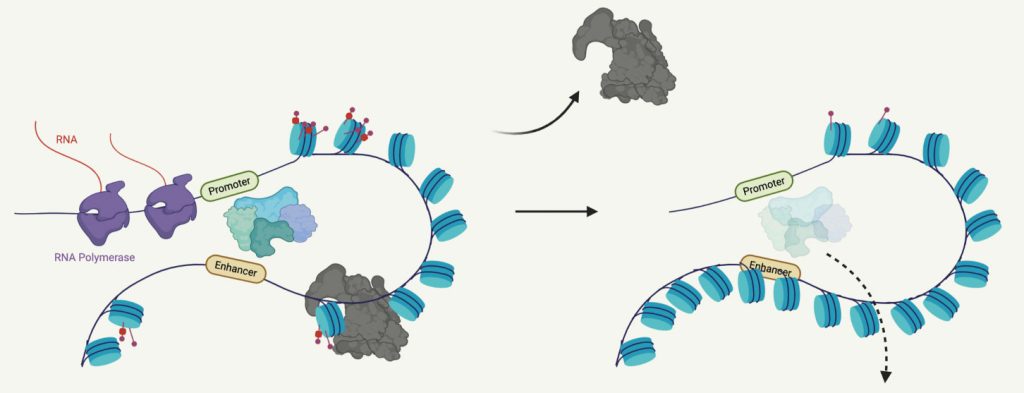
We showed that the cBAF subunit ARID1A regulates chromatin accessibility at enhancers, where it is also essential for maintaining transcription factor binding, active histone modifications and enhancer RNA transcription. Interestingly, promoters are relatively resistant to cBAF disruption. Nevertheless, loss of enhancer architecture in ARID1A knockout cells disrupts gene expression. Further, we found that cBAF is critical for the activation of de novo enhancers and gene expression following rapid retargeting in response to inflammatory stimulation in CD8 T cells and macrophages. ncBAF complexes function cooperatively with cBAF at enhancers; however, ncBAF also controls specific gene programs through select transcription factors. We are continuing to examine the role of BAF complex variants in inflammatory stimulation using genetic and chemical perturbation. Our goal is to build a network defining how these complexes cooperate to regulate transcriptional programs relating to inflammation and immune cell function.
BAF subunit mutations in cancer and therapeutic response
Created in BioRender.
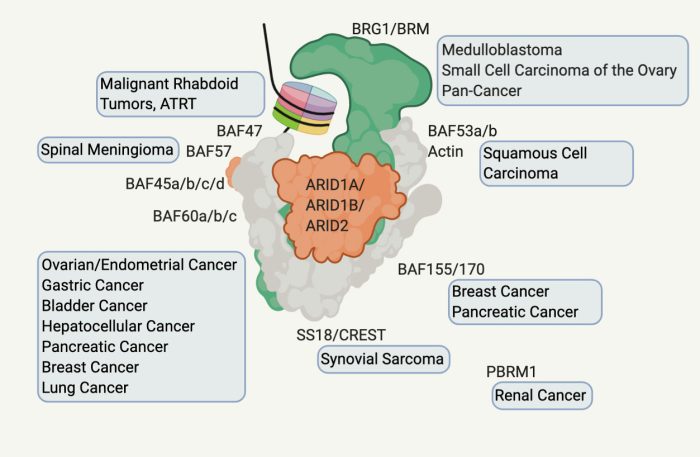
Our analysis of exome sequencing of human cancers demonstrated that loss-of-function mutations in BAF subunits are present in ~20% of all human cancers. In particular, ARID1A is mutated in 10-60% of gynecological and gastrointestinal malignancies. The recent recognition of the high frequency of mutations in ARID1A and other BAF subunits has prompted great interest in understanding the role of these subunits in tumor suppression and therapeutic response.
In cancer patients, ARID1A mutations are significantly enriched among immune checkpoint inhibitor (ICI) responders in many tumor types. We found that genetic deletion or chemical inhibition of ARID1A leads to DNA replication stress and release of single-stranded DNA into the cytosol, which primes interferon-response genes and tumor immunity through a cGAS/STING-dependent mechanism. We further demonstrated that type I interferon is required for the enhanced responsiveness of ARID1A-deficient tumors to ICI immunotherapy in syngeneic mouse models. This activity reflects a function for BAF complexes in the resolution of R loops through an as-yet-undefined mechanism. In related work, we are collaborating with Dr. Gregory Botta (UCSD Moores Cancer Center) to analyze specimens of an ongoing clinical trial of BAF mutant pancreas cancer treated with ICI following spectacular initial results in this normally ICI-unresponsive tumor type. The goal of this work is to determine if ARID1A mutations could serve as biomarkers for therapeutic treatment.
BAF Complex Regulation of Inflammation and Tumor Immunity
Created in BioRender.
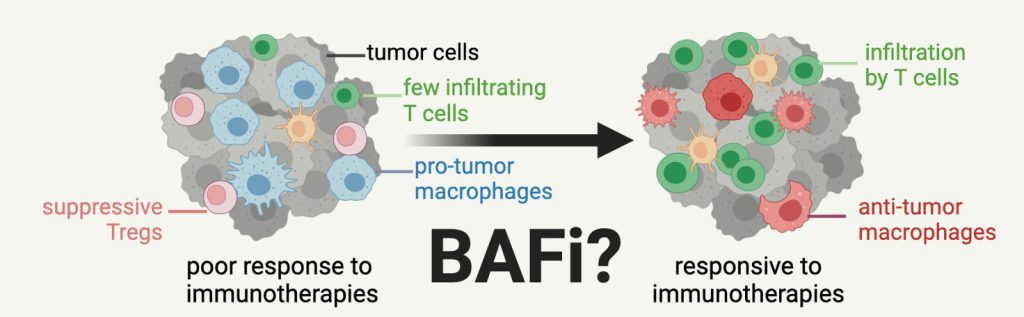
Our work suggests that BAF chemical inhibitors may be effective at sensitizing tumors to ICIs. However, due to their pervasive expression in all cell types, use of BAF inhibitors in cancer therapy could have undesired effects on tumor infiltrating lymphocytes or otherwise healthy tissues. Counterintuitively, our data from immune cells including macrophages, CD8 T cells, and Tregs suggest that BAF complex inhibition is beneficial for controlling immune cell function in cancer and inflammation. Through conditional deletion, chemical inhibition, and epigenomics, we are mapping the activity and function of BAF complexes in immune cells from tumors and inflammatory diseases. Our goal is to determine if BAF inhibitors could be leveraged to toggle inflammatory states for therapeutic purposes in cancer and inflammation.